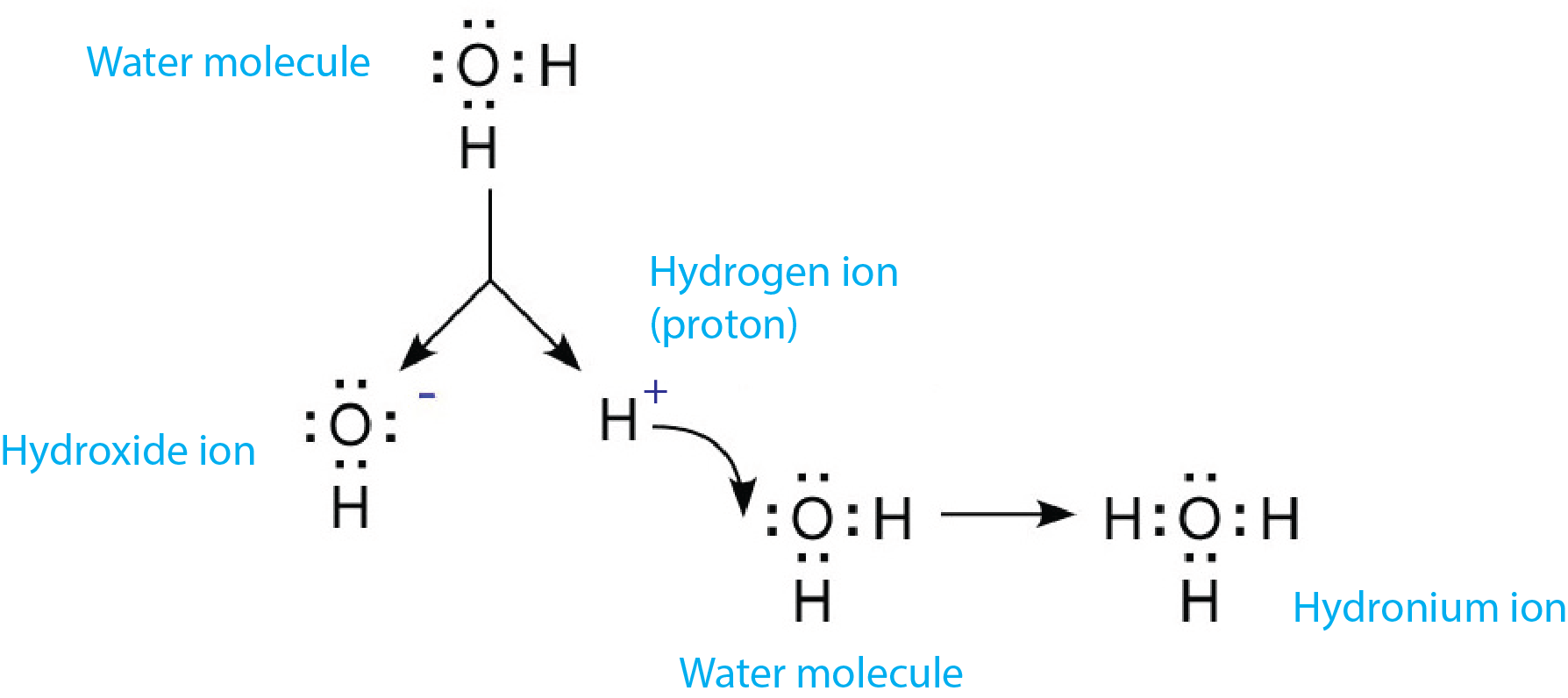
How Does The Hydronium Ion Form
How Does The Hydronium Ion Form - Web when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron is nothing but a proton!). The hydronium ion has a trigonal pyramidal geometry and is composed of 3 hydrogen atoms and 1 oxygen atom. If not, what information do you need in order to calculate it? Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. Thus, in aqueous solution only can a substance dissociate and form hydronium ions and be classified as an acid. 0 + (+1) = +1, i.e. Web $\begingroup$ about the charge: Web hydronium ion at lower ph the hydronium ions (h3o+) available in water protonates the functional groups of melanin and at ph greater than 4, there is a dip in the concentration of hydronium ions leading to the development of negative charge in the functional groups of. It does not matter where exactly the protons and electrons are, the net charge will.
Chapter 5 / lesson 24. Web $\begingroup$ about the charge: Learn about the hydronium cation, which has the chemical formula h3o+. Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Brønsted acids release one or more of their protons (hydrogen ions), which combine with water molecules. Hydronium is the key to calculating whether a solution is acidic or basic. The h 3 o + ion is variously called the hydroxonium ion, the hydronium ion or the oxonium ion. If not, what information do you need in order to calculate it? In an introductory chemistry course (such as gcse), whenever you have talked about. Web it can also be formed by the combination of a h + ion with an h2o molecule.
Web when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron is nothing but a proton!). Web how does the hydronium ion form all acidic aqueous solutions contain protonated water, known commonly as the hydronium ion (h3o+). In an introductory chemistry course (such as gcse), whenever you have talked about. Web in aqueous solution of acids, acid dissociate to form hydrogen ions and combines with water molecules to form hydronium ion (h3o+). Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be charged as well. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. The hydronium ion has a trigonal pyramidal geometry and is composed of 3 hydrogen atoms and 1 oxygen atom. It does not matter where exactly the protons and electrons are, the net charge will. Brønsted acids release one or more of their protons (hydrogen ions), which combine with water molecules. Web $\begingroup$ about the charge:
How Ammonium Ion is formed Formation of Ammonium Ion Hydronium Ion
Web in aqueous solution of acids, acid dissociate to form hydrogen ions and combines with water molecules to form hydronium ion (h3o+). Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Web hydrodium ions are the combination of a water molecule and a hydrogen ion,.
Hydronium Ion or Oxonium
In an introductory chemistry course (such as gcse), whenever you have talked about. Web it can also be formed by the combination of a h + ion with an h2o molecule. If not, what information do you need in order to calculate it? It does not matter where exactly the protons and electrons are, the net charge will. Water is.
Hydronium Ion l Rucete Chemistry In a Nutshell
Web the hydrogen's electron is left behind on the chlorine to form a negative chloride ion. Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be charged as well. In an introductory chemistry course (such as gcse), whenever you have talked about. Web the thing is an acid is any.
Hydronium Ion Easy Science Easy science, Water molecule, Molecules
Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. Thus, in aqueous solution only can a substance dissociate and form hydronium ions and be classified as an acid. Web $\begingroup$ about the charge: Hydronium is the key to calculating whether a solution is acidic or basic. Web in aqueous.
Hydronium Ion Definition & Formula Video & Lesson Transcript
Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Understand the definition of hydronium ion and the. Web when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron is nothing but.
Hydrogen ion Wikipedia
In an introductory chemistry course (such as gcse), whenever you have talked about. Web it can also be formed by the combination of a h + ion with an h2o molecule. However, h+ will not stay a free ion, and instead immediately combines with a nearby water molecule. Web hydrodium ions are the combination of a water molecule and a.
Question Video Calculating the Hydronium Ion Concentration of a
Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be charged as well. Web when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron is nothing but a proton!). The h 3 o + ion is.
Multimedia pH and Color Change Chapter 6, Lesson 8 Middle School
Web when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron is nothing but a proton!). In an introductory chemistry course (such as gcse), whenever you have talked about. Brønsted acids release one or more of their protons (hydrogen ions), which combine with water molecules. Web.
Hydronium Alchetron, The Free Social Encyclopedia
Web when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron is nothing but a proton!). Understand the definition of hydronium ion and the. Brønsted acids release one or more of their protons (hydrogen ions), which combine with water molecules. Web the hydrogen's electron is left.
H3O+, l'ion hydronium définition et explications
In an introductory chemistry course (such as gcse), whenever you have talked about. The h 3 o + ion is variously called the hydroxonium ion, the hydronium ion or the oxonium ion. Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. However, h+ will not.
However, H+ Will Not Stay A Free Ion, And Instead Immediately Combines With A Nearby Water Molecule.
0 + (+1) = +1, i.e. Chapter 5 / lesson 24. Brønsted acids release one or more of their protons (hydrogen ions), which combine with water molecules. Web $\begingroup$ about the charge:
Web When A Protic Acid Is Added To Water, It Releases A Hydrogen Ion (H+) (Also Called Aproton Because A Hydrogen Atom Missing One Electron Is Nothing But A Proton!).
In an introductory chemistry course (such as gcse), whenever you have talked about. Web hydronium ion at lower ph the hydronium ions (h3o+) available in water protonates the functional groups of melanin and at ph greater than 4, there is a dip in the concentration of hydronium ions leading to the development of negative charge in the functional groups of. Learn about the hydronium cation, which has the chemical formula h3o+. The hydronium ion has a trigonal pyramidal geometry and is composed of 3 hydrogen atoms and 1 oxygen atom.
Web It Can Also Be Formed By The Combination Of A H + Ion With An H2O Molecule.
Hydronium is the key to calculating whether a solution is acidic or basic. It does not matter where exactly the protons and electrons are, the net charge will. Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Calculate the hydronium ion concentration of human blood.
If Not, What Information Do You Need In Order To Calculate It?
The h 3 o + ion is variously called the hydroxonium ion, the hydronium ion or the oxonium ion. Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be charged as well. Understand the definition of hydronium ion and the. Web how does the hydronium ion form all acidic aqueous solutions contain protonated water, known commonly as the hydronium ion (h3o+).