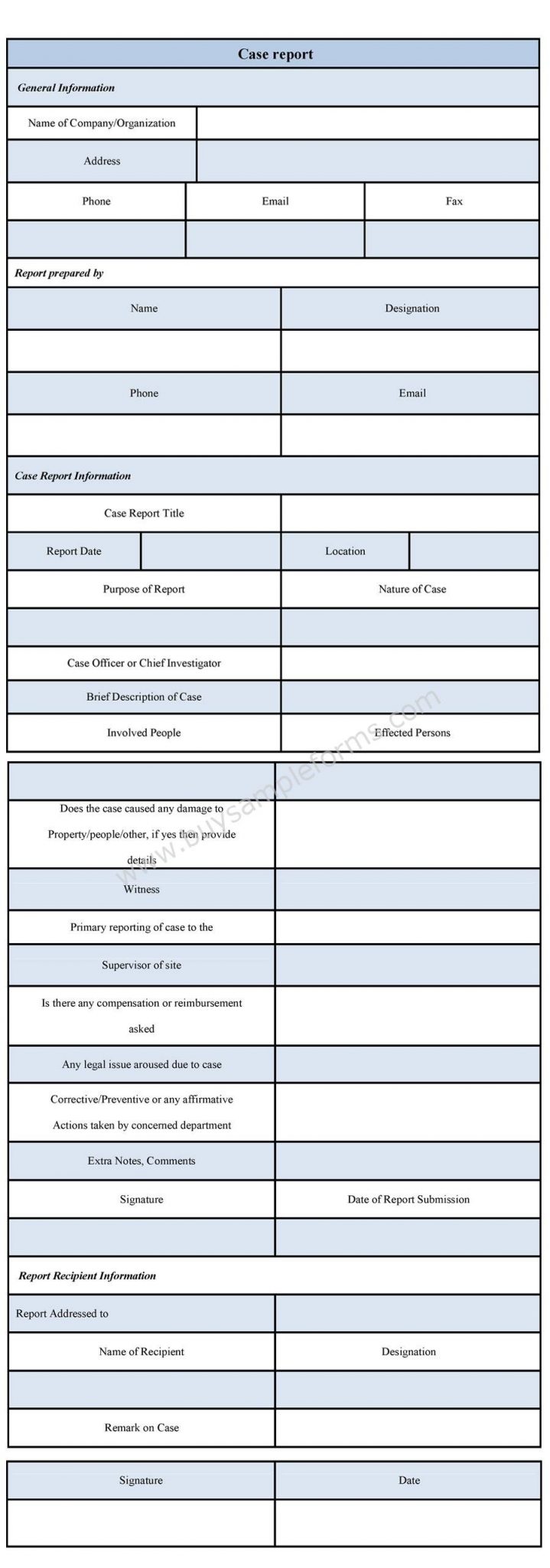
Case Report Form
Case Report Form - Case report form (crf) is a specialized document in clinical research. All data on each patient participating in a clinical trial are held and/or documented in the crf. Most of the time, participants in clinical research are not even aware of crfs. Its development represents a significant part of the clinical trial and can affect study success. Case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of a study. Though paper crfs are still used largely, use of electronic crfs (ecrfs) are gaining popularity due to the advantages they offer such as improved data. Web case report forms (or crfs, for short) are an integral component of clinical trials. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Each clinical trial participant has a. [1] the case report form is the tool used by the sponsor of the clinical trial to collect data from each participating patient.
But they are a key component for recording the data in a clinical trial. Though paper crfs are still used largely, use of electronic crfs (ecrfs) are gaining popularity due to the advantages they offer such as improved data. The cdash standards identify those elements that should be captured on a case report form (crf). Its development represents a significant part of the clinical trial and can affect study success. Case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of a study. These templates are consistent with the fda cdash (clinical data acquisition standards harmonization) standards. Each clinical trial participant has a. Web case report forms (or crfs, for short) are an integral component of clinical trials. Most of the time, participants in clinical research are not even aware of crfs. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine.
Web what is a case report form? Research data is ultimately submitted to the sponsor and/or analyzed by the emory investigator by either paper case report forms (crfs) or. [1] the case report form is the tool used by the sponsor of the clinical trial to collect data from each participating patient. Case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of a study. Web a case report form (crf) is designed to collect the patient data in a clinical trial; Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. But they are a key component for recording the data in a clinical trial. It should be study protocol driven, robust in content and have material to collect the study specific data. Case report form (crf) is a specialized document in clinical research. Web case classification and identification what is the current status of this person?
What Is a Case Report Form? [ Importance, Tips, Samples ]
Epix notification of travelers meets clinical criteria and epidemiologic evidence with no confirmatory lab testing* Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web what is a case report form? Web case report forms (or crfs, for short) are an integral component of clinical trials. These templates are consistent.
FREE 15+ Case Report Forms in PDF MS Word
Web case report forms (or crfs, for short) are an integral component of clinical trials. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine. [1] the case report form is the tool used by the sponsor of the clinical trial to collect.
Free 15+ Case Report Forms In Pdf Ms Word Inside Case Report Form
Web a case report form (crf) is designed to collect the patient data in a clinical trial; It should be study protocol driven, robust in content and have material to collect the study specific data. These templates are consistent with the fda cdash (clinical data acquisition standards harmonization) standards. Case report forms (crfs) are arguably the most important documentation in.
FREE 15+ Case Report Forms in PDF MS Word
Case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of a study. Web what is a case report form? These templates are consistent with the fda cdash (clinical data acquisition standards harmonization) standards. All data on each patient participating in a.
Case Report Form Case Report Format
Epix notification of travelers meets clinical criteria and epidemiologic evidence with no confirmatory lab testing* Case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of a study. The cdash standards identify those elements that should be captured on a case report.
10+ Case Report Templates PDF, Word, Pages Free & Premium Templates
Research data is ultimately submitted to the sponsor and/or analyzed by the emory investigator by either paper case report forms (crfs) or. Though paper crfs are still used largely, use of electronic crfs (ecrfs) are gaining popularity due to the advantages they offer such as improved data. Epix notification of travelers meets clinical criteria and epidemiologic evidence with no confirmatory.
FREE 15+ Case Report Forms in PDF MS Word
Epix notification of travelers meets clinical criteria and epidemiologic evidence with no confirmatory lab testing* Web case classification and identification what is the current status of this person? It should be study protocol driven, robust in content and have material to collect the study specific data. A case report form is an essential tool for clinical research, although it has.
Blank Case Report Form RIAT Support Center
Web case classification and identification what is the current status of this person? All data on each patient participating in a clinical trial are held and/or documented in the crf. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine. Case report forms.
Blank Case Report Form RIAT Support Center
Web a case report form (crf) is designed to collect the patient data in a clinical trial; Web case report forms (or crfs, for short) are an integral component of clinical trials. But they are a key component for recording the data in a clinical trial. Though paper crfs are still used largely, use of electronic crfs (ecrfs) are gaining.
FREE 15+ Case Report Forms in PDF MS Word
Case report form (crf) is a specialized document in clinical research. Epix notification of travelers meets clinical criteria and epidemiologic evidence with no confirmatory lab testing* Web what is a case report form? The crf facilitates complete and standardized data collection that promotes efficient processing, analysis, and reporting of information, as well as exchange of data across. The cdash standards.
Web What Is A Case Report Form?
The crf facilitates complete and standardized data collection that promotes efficient processing, analysis, and reporting of information, as well as exchange of data across. Web a case report form (crf) is designed to collect the patient data in a clinical trial; [ 1] site personnel capture the subject's data on the crf, which is collected during their participation in a clinical trial. Research data is ultimately submitted to the sponsor and/or analyzed by the emory investigator by either paper case report forms (crfs) or.
These Templates Are Consistent With The Fda Cdash (Clinical Data Acquisition Standards Harmonization) Standards.
A case report form is an essential tool for clinical research, although it has been extended for use in other areas of medicine. [1] the case report form is the tool used by the sponsor of the clinical trial to collect data from each participating patient. Epix notification of travelers meets clinical criteria and epidemiologic evidence with no confirmatory lab testing* Web case classification and identification what is the current status of this person?
Its Development Represents A Significant Part Of The Clinical Trial And Can Affect Study Success.
Web case report forms (or crfs, for short) are an integral component of clinical trials. All data on each patient participating in a clinical trial are held and/or documented in the crf. Most of the time, participants in clinical research are not even aware of crfs. Case report form (crf) is a specialized document in clinical research.
The Cdash Standards Identify Those Elements That Should Be Captured On A Case Report Form (Crf).
Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Case report forms (crfs) are arguably the most important documentation in a clinical trial since they are the last point of data entry, which ultimately influences the outcome of a study. Each clinical trial participant has a. Though paper crfs are still used largely, use of electronic crfs (ecrfs) are gaining popularity due to the advantages they offer such as improved data.
![What Is a Case Report Form? [ Importance, Tips, Samples ]](https://images.sampleforms.com/wp-content/uploads/2018/02/Confidential-Case-Report-Form-in-PDF-1.jpg)