Which Of The Following Would Form An Electrolyte Solution
Which Of The Following Would Form An Electrolyte Solution - Choose all that are correct. Web you'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web which of the following solutes would form electrolyte solutions when dissolved in water? Web substances that dissolve in water to yield ions are called electrolytes. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web when some substances are dissolved in water, they undergo either a physical or a chemical change that yields ions in solution. Electrolytes found in your body include: The most important electrolytes found in the highest amounts in the body are sodium,. Electrolysis is the decomposition of an electrolyte by an electric current. Solutions of electrolytes contain ions that permit the.
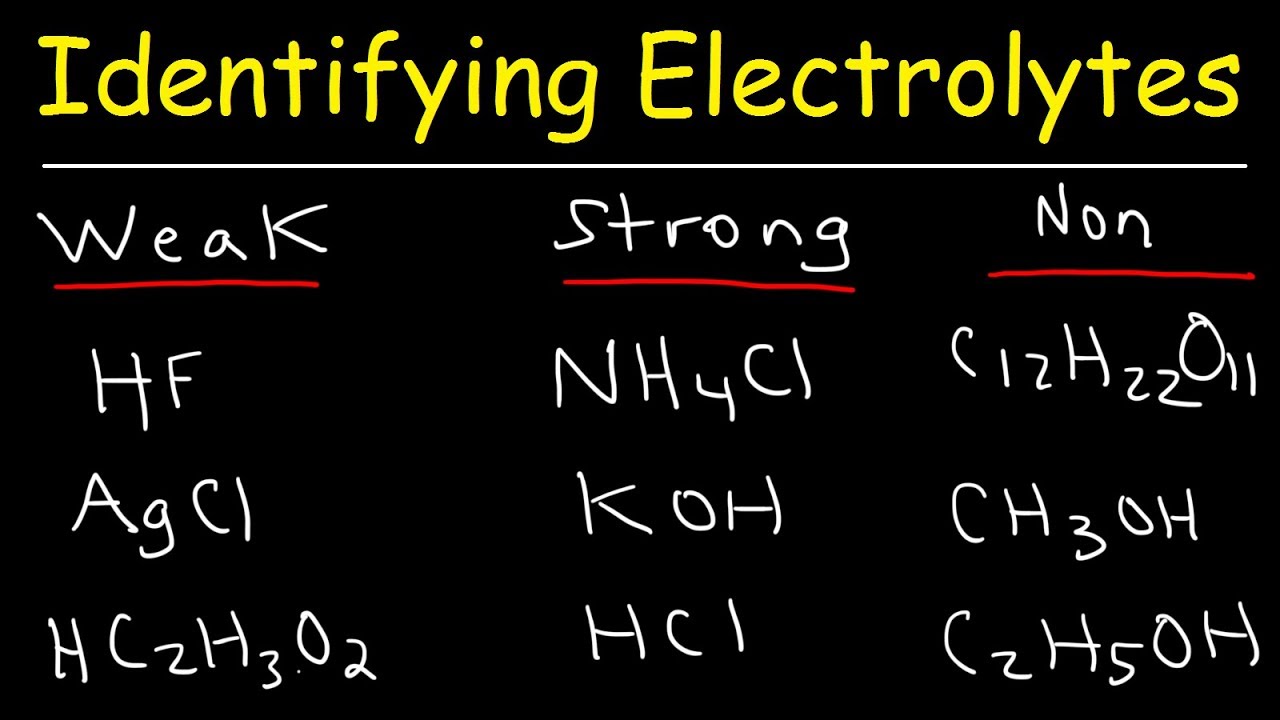
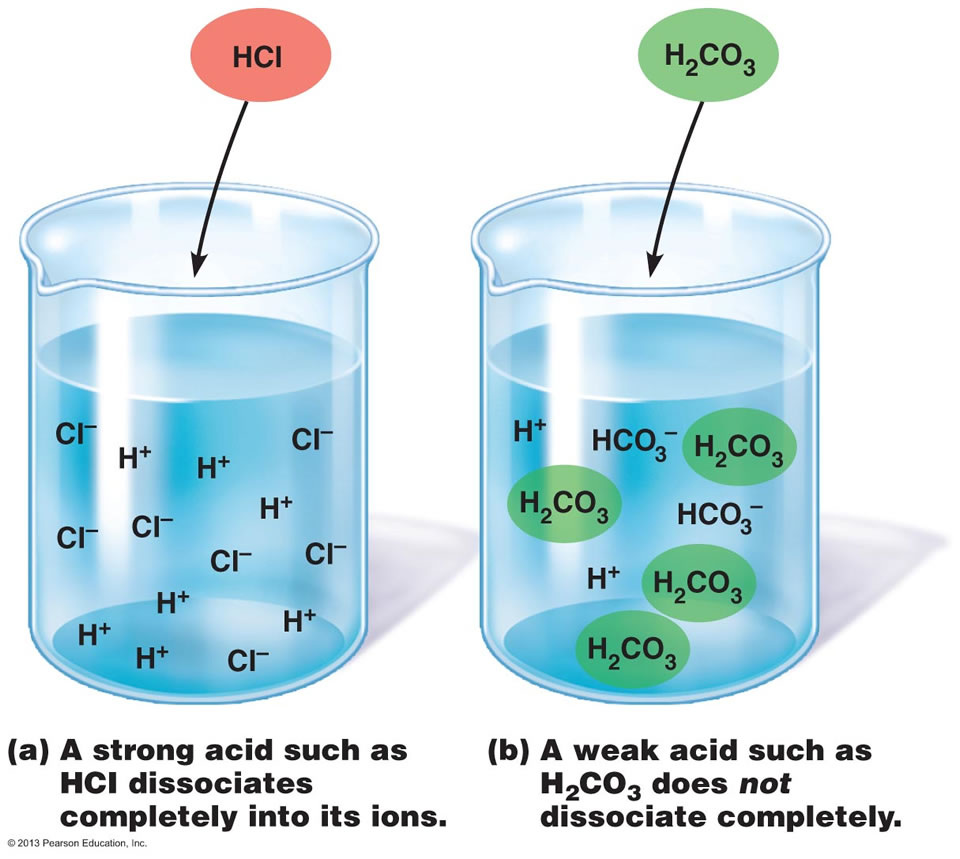
Web when some substances are dissolved in water, they undergo either a physical or a chemical change that yields ions in solution. Web beverages like coconut water and fruit smoothies also provide electrolytes. Web important electrolytes other than sodium and chloride include potassium, calcium, bicarbonate and phosphate. Which of the following would form an electrolyte solution? Web when some substances are dissolved in water, they undergo either a physical or a chemical change that yields ions in solution. Web strong electrolytes completely ionize when dissolved, and no neutral molecules are formed in solution. These substances constitute an important class of. Web verified answered this week 1 of 3 salt in water would form an electrolyte solution. Choose all that are correct. Solutions of electrolytes contain ions that permit the.
Web strong electrolytes completely ionize when dissolved, and no neutral molecules are formed in solution. These substances constitute an important class of. Web beverages like coconut water and fruit smoothies also provide electrolytes. Solutions of electrolytes contain ions that permit the. Web the molten or dissolved substance is called the electrolyte. It is used to extract reactive. Electrolyte solutions are composed of ions dissolved in water allowing it to conduct. Web important electrolytes other than sodium and chloride include potassium, calcium, bicarbonate and phosphate. Which of the following would form an electrolyte solution? Web substances that dissolve in water to yield ions are called electrolytes.
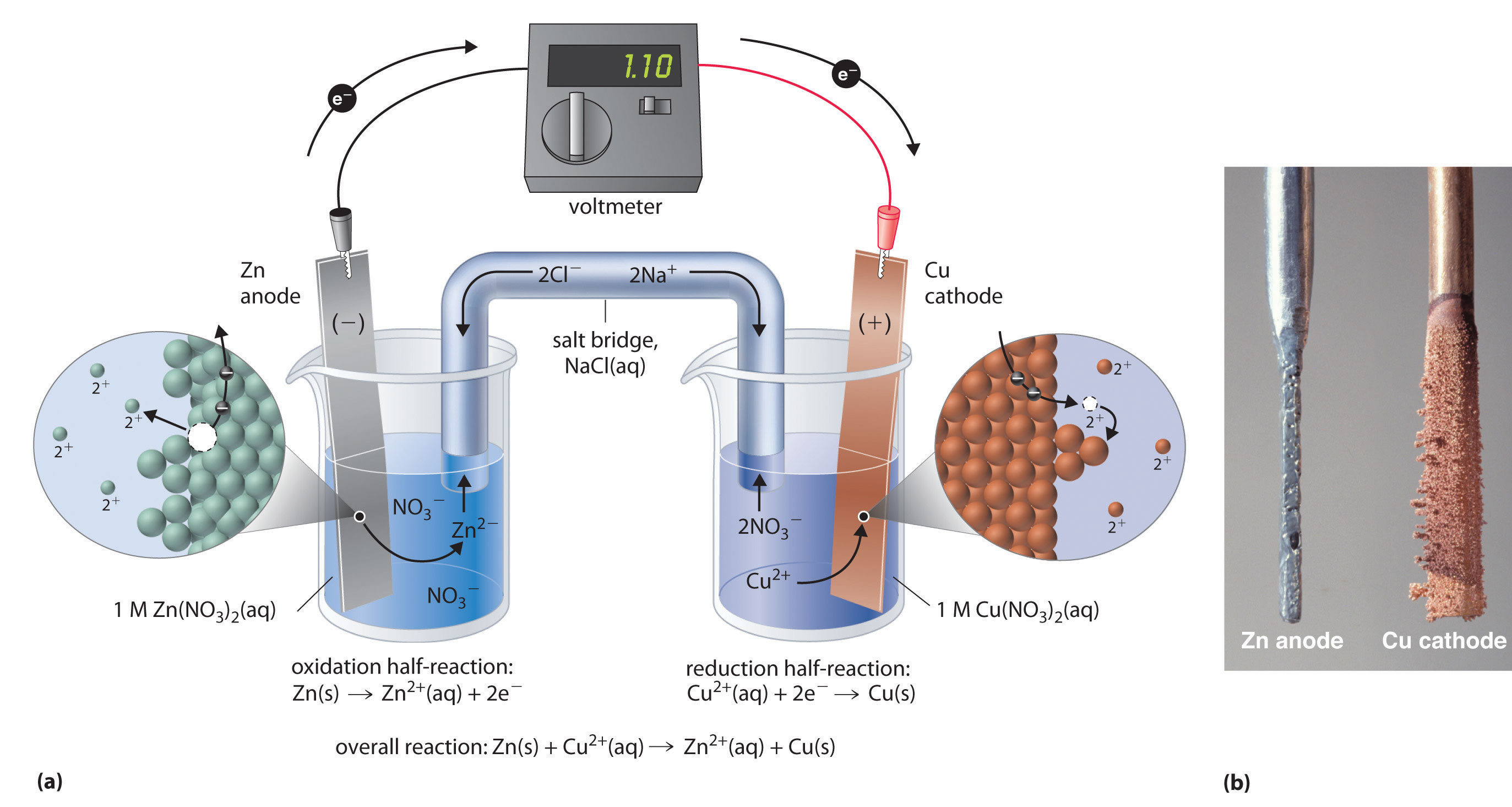
Daniell Cell Study Material for IIT JEE askIITians
Web strong electrolytes completely ionize when dissolved, and no neutral molecules are formed in solution. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web the molten or dissolved substance is called the electrolyte. Web substances that dissolve in water to yield ions are called electrolytes. It is used to extract reactive.
Solved 1. Which of the following substances is a strong
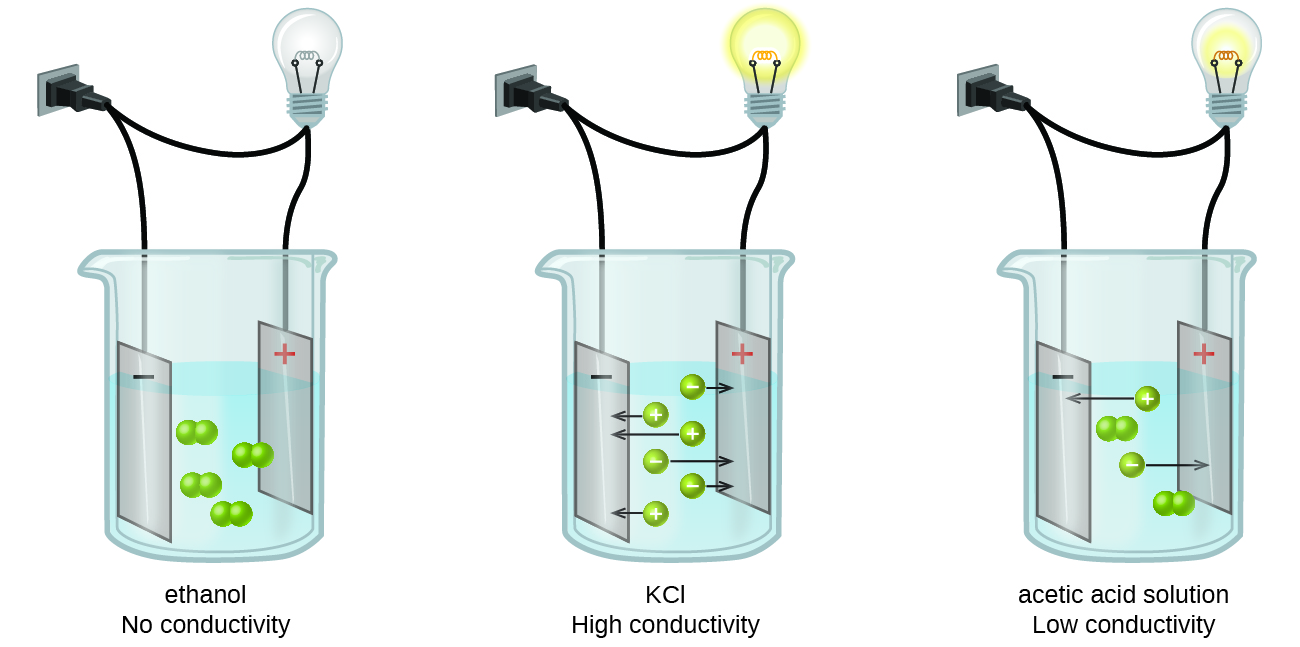
Holding 100ml of water (ebkare)________________2. Which of the following would form an electrolyte solution? Electrolytes found in your body include: You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Solutions of nonelectrolytes such as ethanol do not contain dissolved ions and cannot conduct electricity.
Electrolytic Cells Video & Lesson Transcript
Web important electrolytes other than sodium and chloride include potassium, calcium, bicarbonate and phosphate. Web beverages like coconut water and fruit smoothies also provide electrolytes. Solutions of nonelectrolytes such as ethanol do not contain dissolved ions and cannot conduct electricity. The most important electrolytes found in the highest amounts in the body are sodium,. Web when some substances are dissolved.
Solved Classify each of the following solutes as a strong
Electrolyte solutions are composed of ions dissolved in water allowing it to conduct. Holding 100ml of water (ebkare)________________2. Web when some substances are dissolved in water, they undergo either a physical or a chemical change that yields ions in solution. Web this problem has been solved! Solutions of electrolytes contain ions that permit the.
Identifying Strong Electrolytes, Weak Electrolytes, and Nonelectrolytes
Solutions of electrolytes contain ions that permit the. Solutions of nonelectrolytes such as ethanol do not contain dissolved ions and cannot conduct electricity. These substances constitute an important class of. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Electrolyte solutions are composed of ions dissolved in water allowing it to conduct.
Question a0b96 + Example
These substances constitute an important class of. Web beverages like coconut water and fruit smoothies also provide electrolytes. Web this problem has been solved! For example, nacl, hno 3, hclo 3, cacl 2 etc. Web substances that dissolve in water to yield ions are called electrolytes.
Electrolytes Chemistry for Majors
Web chemistry chemistry questions and answers di question 4 0.75 pts which of the following would not form an electrolyte solution when added to water? Measuring 27 ml of liquid (daudgtear ldnreiyc)________________3. Web verified answered this week 1 of 3 salt in water would form an electrolyte solution. Which of the following would form an electrolyte solution? Web substances that.
Two halfcells in a galvanic cell consist of one iron (Fe(s)) electrode
Web the current view of electrolyte solutions is that, in water at normal temperatures, the salts of strong acids and strong bases are completely dissociated into ions at all. For example, nacl, hno 3, hclo 3, cacl 2 etc. These substances constitute an important class of. Electrolyte solutions are composed of ions dissolved in water allowing it to conduct. Web.
Which Of The Following Diagrams Represent A Weak Acid Dissolved In
Web which of the following solutes would form electrolyte solutions when dissolved in water? Web chemistry chemistry questions and answers di question 4 0.75 pts which of the following would not form an electrolyte solution when added to water? Solutions of electrolytes contain ions that permit the. For example, nacl, hno 3, hclo 3, cacl 2 etc. Electrolyte solutions are.
4.1 Electrolytes Chemistry LibreTexts
Web when these minerals dissolve in a fluid, they form electrolytes — positive or negative ions in metabolic processes. Electrolysis is the decomposition of an electrolyte by an electric current. Web which of the following solutes would form electrolyte solutions when dissolved in water? Group of answer choices sulfuric acid, h2so4 ethanol,. The most important electrolytes found in the highest.
Measuring 27 Ml Of Liquid (Daudgtear Ldnreiyc)________________3.
These substances constitute an important class of. Web when some substances are dissolved in water, they undergo either a physical or a chemical change that yields ions in solution. Which of the following would form an electrolyte solution? Which of the following would form an electrolyte solution when.
Solutions Of Nonelectrolytes Such As Ethanol Do Not Contain Dissolved Ions And Cannot Conduct Electricity.
Electrolysis is the decomposition of an electrolyte by an electric current. For example, nacl, hno 3, hclo 3, cacl 2 etc. Electrolyte solutions are composed of ions dissolved in water allowing it to conduct. Web this problem has been solved!
Choose All That Are Correct.
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web strong electrolytes completely ionize when dissolved, and no neutral molecules are formed in solution. Web which of the following solutes would form electrolyte solutions when dissolved in water? Web when these minerals dissolve in a fluid, they form electrolytes — positive or negative ions in metabolic processes.
Web Substances That Dissolve In Water To Yield Ions Are Called Electrolytes.
Web the molten or dissolved substance is called the electrolyte. Web the current view of electrolyte solutions is that, in water at normal temperatures, the salts of strong acids and strong bases are completely dissociated into ions at all. Electrolytes may be covalent compounds that chemically react with water to produce ions (for example, acids. These substances constitute an important class of.