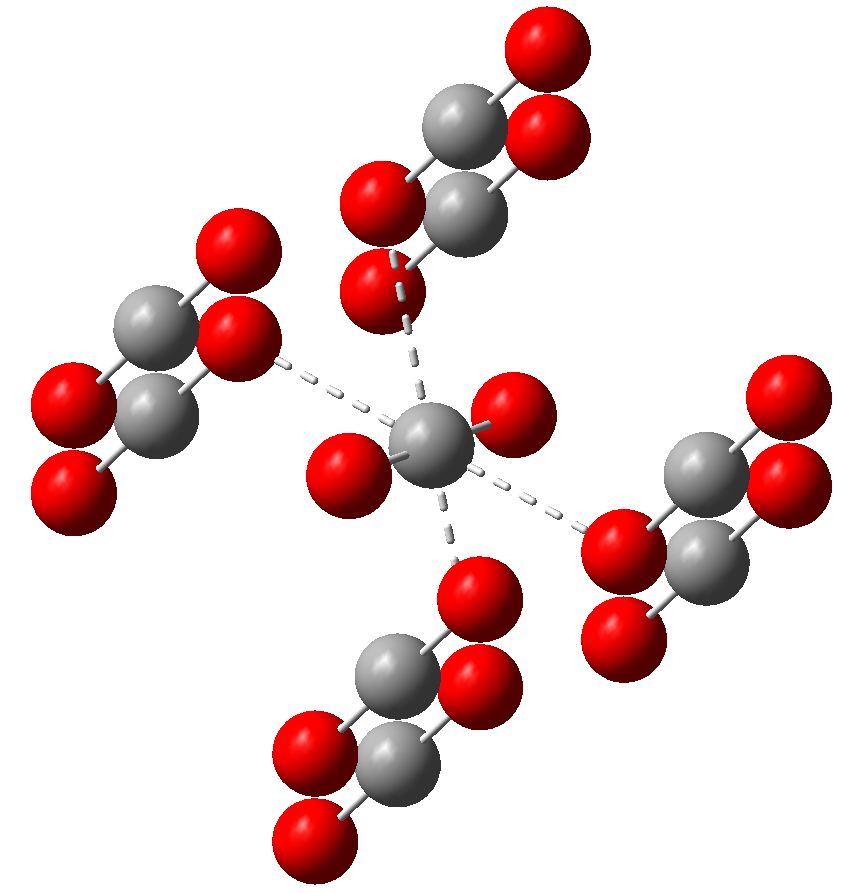
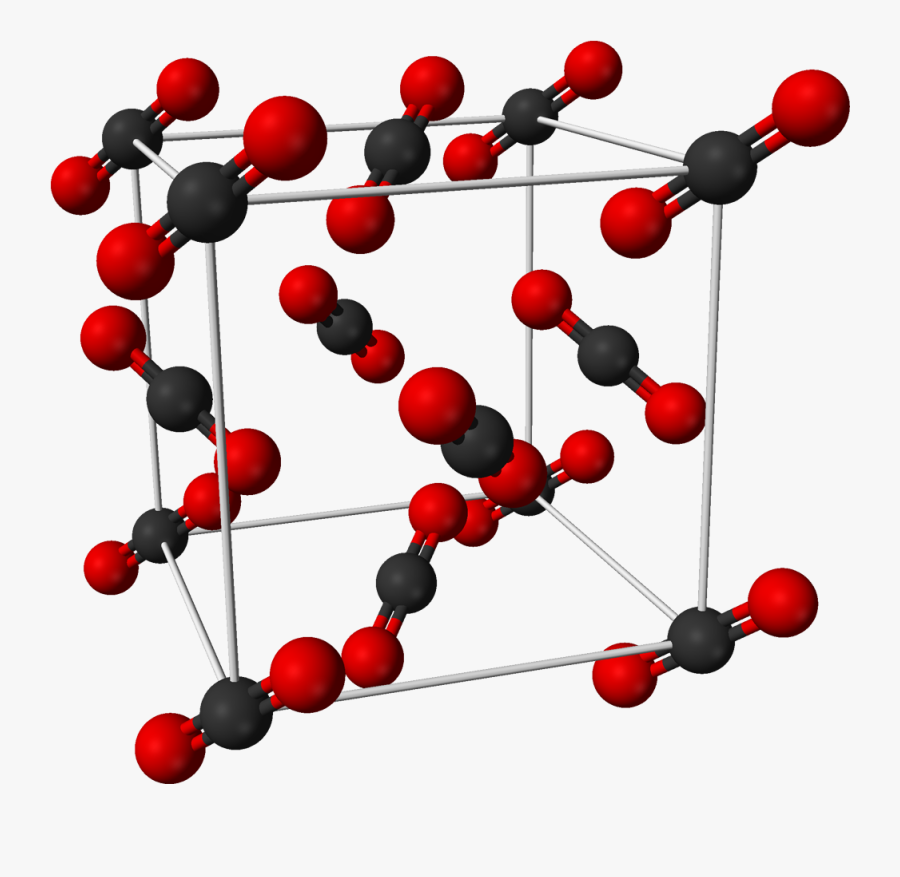
Solid Form Of Carbon Dioxide
Solid Form Of Carbon Dioxide - In typical co 2 capture processes, the co 2 gas is pressurised to a liquid, which is then transported to a. Ou chemical safety data (no longer updated) more details −109.3 °f) at earth atmospheric pressure — that is, it transitions directly from solid to. Solid form is utilized as dry ice.] It's called “dry” ice because instead of melting into a liquid like regular ice melts into water, dry ice sublimes directly into the gaseous state of carbon dioxide. Web carbon dioxide (chemical formula: Carbon dioxide is a gas at room temperature,. It is used to preserve food that stores in cool temperature without any mechanical tool of cooling like ice cream. Freezer burn results from the sublimation of ice into water vapor. Cooled co 2 in solid form is called dry ice.
Web dry ice is the solid form of carbon dioxide , the molecule that animals breathe out when we exhale and plants take in when they do photosynthesis. Web wear insulating gloves if handling solid carbon dioxide.if working with carbon dioxide in confined spaces where the concentration of gas may build up, ensure adequate ventilation. Solid carbon dioxide is always below −78.5 °c (−109.3 °f) at regular atmospheric pressure, regardless of the air temperature. Carbon dioxide is a gas at room temperature,. Web science news from research organizations decarbonization tech instantly converts carbon dioxide to solid carbon date: Find out about the process of sublimation, the uses. Dry ice is the solid form of carbon dioxide. Freezer burn results from the sublimation of ice into water vapor. Shipped as a liquefied compressed gas. The chemical or molecular formula for carbon dioxide is co 2.
The chemical or molecular formula for carbon dioxide is co 2. Web carbon dioxide forms a weak acid, called carbonic acid (h 2 co 3), when dissolved in water. They are currently the largest energy sources for electricity production in conventional thermal power plants. People and animals release carbon dioxide when they breathe out. At room temperature and pressure, it sublimates into carbon dioxide vapor. It is relatively nontoxic and noncombustible, but it is heavier than air and may asphyxiate by the displacement of air. Web answer (1 of 6): It is also used in laboratories for reacting chemicals that require very low temperature. Solid co 2 sublimes at 194.65 k (−78.5 °c; Web solid carbons such as coal and biomass are cheap fuels with high energy density in volume.
Carbon dioxide wikidoc
Web wear insulating gloves if handling solid carbon dioxide.if working with carbon dioxide in confined spaces where the concentration of gas may build up, ensure adequate ventilation. Web carbon dioxide, solid chemical identifiers | hazards | response recommendations | physical properties | regulatory information | alternate chemical names chemical identifiers Solid, frozen carbon dioxide is called dry ice. It is.
CO2solid
People and animals release carbon dioxide when they breathe out. They are widely distributed all over the world. In this reaction, solid carbon latches onto one of the oxygen atoms in carbon dioxide gas,. Web dry ice is the solid form of carbon dioxide , the molecule that animals breathe out when we exhale and plants take in when they.
Media Portfolio
Solid carbon dioxide is always below −78.5 °c (−109.3 °f) at regular atmospheric pressure, regardless of the air temperature. Web dry ice is the solid form of carbon dioxide, a molecule that is found as a gas in the air. It is absorbed by plants in photosynthesis. Web dry ice is the solid form of carbon dioxide (co 2), a.
Researchers discover a way to tease oxygen molecules from carbon dioxide
It is also used in laboratories for reacting chemicals that require very low temperature. Carbon dioxide is the most abundant gas in the atmospheres of mars and venus. They are widely distributed all over the world. Web answer (1 of 10): Web solid carbons such as coal and biomass are cheap fuels with high energy density in volume.
How Much does Dry Ice Cost
In typical co 2 capture processes, the co 2 gas is pressurised to a liquid, which is then transported to a. It is formed by combustion and by biological processes. The chemical structure is centrosymmetric and linear, so carbon dioxide has no electric dipole. The central carbon atom is joined to two oxygen atoms by covalent double bonds. Web carbonic.
A view to a kill in the morning Carbon dioxide Scientific American
Web answer (1 of 10): Web a new carbon capture process has been developed that turns carbon dioxide gas into solid carbon that is easier to store. The chemical or molecular formula for carbon dioxide is co 2. Shipped as a liquefied compressed gas. Web dry ice is the solid form of carbon dioxide , the molecule that animals breathe.
Illustrated Glossary of Organic Chemistry Carbon dioxide
Ou chemical safety data (no longer updated) more details Web dry ice is the solid form of carbon dioxide (co 2), a molecule consisting of a single carbon atom bonded to two oxygen atoms. Web carbon dioxide, represented by the chemical formula co2, is a colorless gas in the atmosphere that is one part carbon and two parts oxygen. Web.
There is More Carbon Dioxide in the Atmosphere Than Ever Before
It is formed by combustion and by biological processes. Solid, frozen carbon dioxide is called dry ice. This is a very used and efficient refrigerant, which when losing cold does not transform into water (like common ice) but turns into gas (since at atmospheric pressure it is gaseous), which tends to minimize microbial contamination in fresh. In typical co 2.
What is the solid form of carbon... Trivia Questions
Solid, frozen carbon dioxide is called dry ice. When a chunk of dry ice is exposed to room temperature air it undergoes sublimation, which means it changes from a solid directly into a gas, without melting into a liquid first. They are currently the largest energy sources for electricity production in conventional thermal power plants. Web carbonic acid gas, dry.
File Carbon Dioxide Unit Solid Carbon Dioxide Structure , Free
Ou chemical safety data (no longer updated) more details It is formed by combustion and by biological processes. Web carbon dioxide (chemical formula: It is used to preserve food that stores in cool temperature without any mechanical tool of cooling like ice cream. People and animals release carbon dioxide when they breathe out.
Web Dry Ice Is The Solid Form Of Carbon Dioxide (Co 2), A Molecule Consisting Of A Single Carbon Atom Bonded To Two Oxygen Atoms.
It's called “dry” ice because instead of melting into a liquid like regular ice melts into water, dry ice sublimes directly into the gaseous state of carbon dioxide. Normal constituent of air (about 300 ppm)]. Solid form is utilized as dry ice.] At room temperature and pressure, it sublimates into carbon dioxide vapor.
Suggest New Co2 Full Form.
Web carbonic acid gas, dry ice [note: They are currently the largest energy sources for electricity production in conventional thermal power plants. Carbon dioxide is a gas at room temperature,. The common name of solid carbon dioxide (co2) is “dry ice”.
Web In Solid Form, It Is Called Dry Ice.
Web sublimation examples dry ice is solid carbon dioxide. It is formed by combustion and by biological processes. Solid carbon dioxide is always below −78.5 °c (−109.3 °f) at regular atmospheric pressure, regardless of the air temperature. Web carbon dioxide, co2, is a colourless and odorless gas.
In This Reaction, Solid Carbon Latches Onto One Of The Oxygen Atoms In Carbon Dioxide Gas,.
Find out about the process of sublimation, the uses. Web carbon dioxide forms a weak acid, called carbonic acid (h 2 co 3), when dissolved in water. When a chunk of dry ice is exposed to room temperature air it undergoes sublimation, which means it changes from a solid directly into a gas, without melting into a liquid first. It is absorbed by plants in photosynthesis.