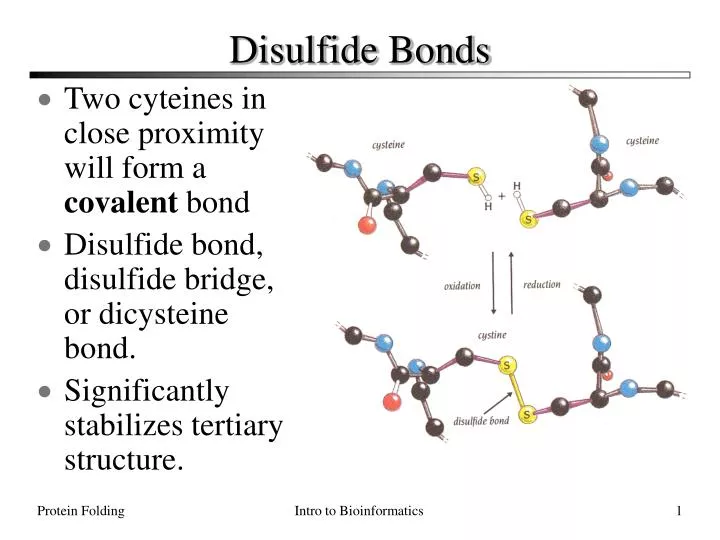
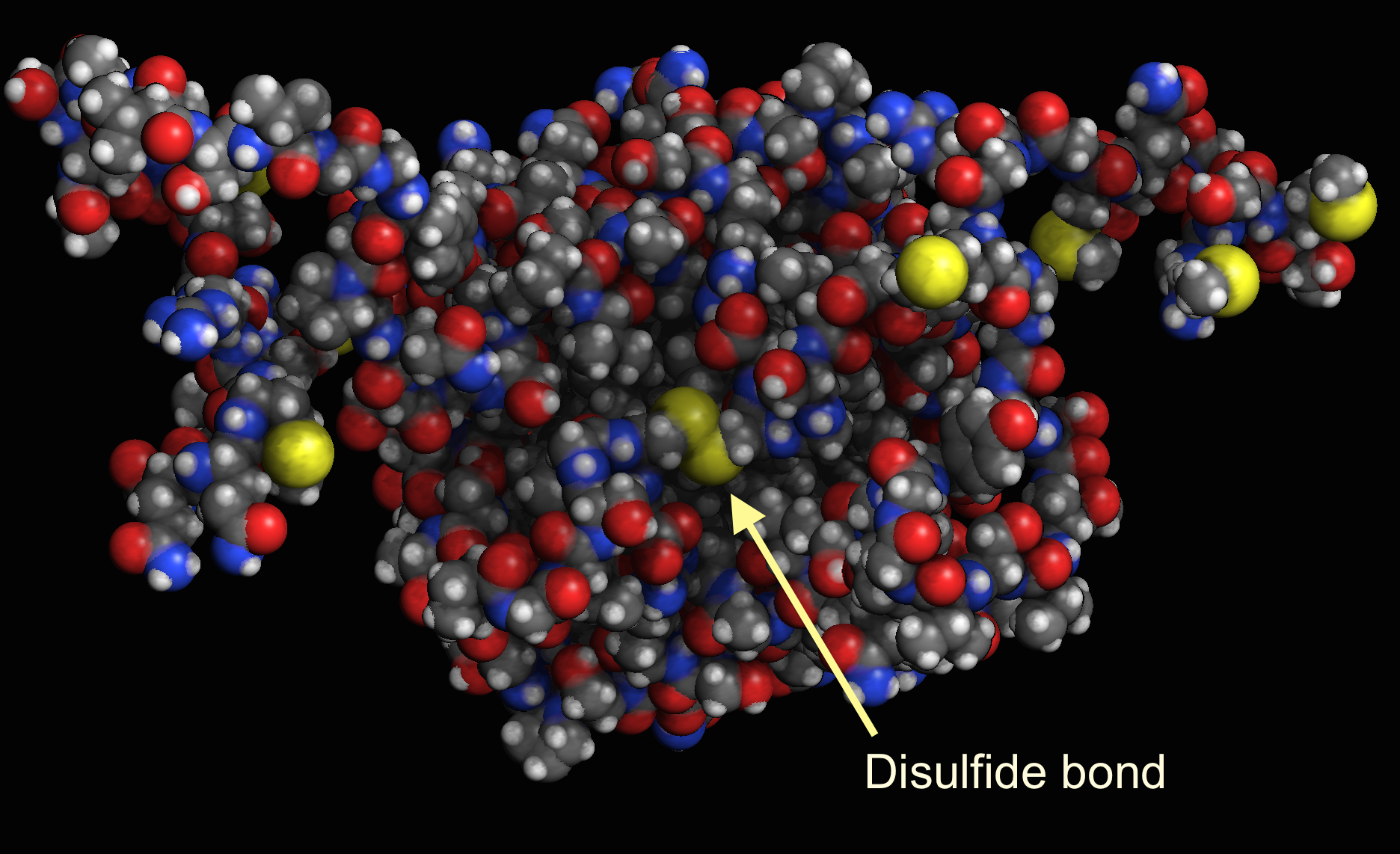
Can Methionine Form Disulfide Bonds
Can Methionine Form Disulfide Bonds - Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so with other cysteine groups. Web is cysteine the only amino acid that can form disulfide bonds? Cysteine residues function in the catalytic cycle of many enzymes, and they can form disulfide bonds that contribute to protein structure. Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. An s− anion from one sulfhydryl group acts as a nucleophile, attacking the side chain of a second cysteine to create a disulfide bond, and in the process releases electrons (reducing equivalents) for transfer. Thus methionine is more hydrophobic, sterically. Web answer (1 of 4): Web methionine residues as endogenous antioxidants in proteins.
Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Web methionine residues as endogenous antioxidants in proteins. An s− anion from one sulfhydryl group acts as a nucleophile, attacking the side chain of a second cysteine to create a disulfide bond, and in the process releases electrons (reducing equivalents) for transfer. Web what can form disulfide bonds? Web answer (1 of 4): Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so with other cysteine groups. Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Thus methionine is more hydrophobic, sterically.
Web what can form disulfide bonds? Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so with other cysteine groups. Cysteine residues function in the catalytic cycle of many enzymes, and they can form disulfide bonds that contribute to protein structure. Thus methionine is more hydrophobic, sterically. Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web is cysteine the only amino acid that can form disulfide bonds? An s− anion from one sulfhydryl group acts as a nucleophile, attacking the side chain of a second cysteine to create a disulfide bond, and in the process releases electrons (reducing equivalents) for transfer. Web methionine residues as endogenous antioxidants in proteins.
The methionine at peptide position 5 alters significantly the
Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Web answer (1 of 4): Web is cysteine the only amino acid that can form disulfide bonds? Cysteine residues function in the catalytic cycle of.
Chapter 2 Protein Structure Chemistry
Cysteine residues function in the catalytic cycle of many enzymes, and they can form disulfide bonds that contribute to protein structure. Web methionine residues as endogenous antioxidants in proteins. Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Disulfide bonds in proteins are formed between the thiol groups of cysteine residues.
Geometry of a disulfide bond. The covalent bond between the sulfur
An s− anion from one sulfhydryl group acts as a nucleophile, attacking the side chain of a second cysteine to create a disulfide bond, and in the process releases electrons (reducing equivalents) for transfer. Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Web cystine is composed of two cysteines linked by.
Mechanisms of cleavage of allosteric disulfide bonds. Disulfide bond
Cysteine residues function in the catalytic cycle of many enzymes, and they can form disulfide bonds that contribute to protein structure. Thus methionine is more hydrophobic, sterically. Web what can form disulfide bonds? Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Cysteine residues disulfide bonds in proteins are formed between the.
Arrangement of disulfide bonds in mature proteins. Download
Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so with other cysteine groups. Cysteine residues function in the catalytic cycle of many enzymes, and they can form disulfide bonds that contribute to protein structure. Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains.
PPT Disulfide Bonds PowerPoint Presentation ID165240
Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so with other cysteine groups. Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web what can form disulfide bonds? Web methionine residues as endogenous antioxidants in proteins. Cysteine.
Along came a spider ScienceBlogs
Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Web methionine residues as endogenous antioxidants in proteins. Web is cysteine the only amino acid that can form disulfide bonds? Web.
Why Can'T Methionine Form Disulfide Bonds? The 8 Top Answers
Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. An s− anion from one sulfhydryl group acts as a nucleophile, attacking the side chain of a second cysteine to create a.
Protein Secretion and the Endoplasmic Reticulum
Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web is cysteine the only amino acid that can form disulfide bonds? An s− anion from one sulfhydryl group acts.
Disulfide bond wikidoc
Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Web is cysteine the only amino acid that can form disulfide bonds? Web answer (1 of 4): Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so.
Web Is Cysteine The Only Amino Acid That Can Form Disulfide Bonds?
Web the cysteine amino acid group is the only amino acid capable of forming disulfide bonds, and thus can only do so with other cysteine groups. Disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding. Cysteine residues function in the catalytic cycle of many enzymes, and they can form disulfide bonds that contribute to protein structure. Cysteine residues disulfide bonds in proteins are formed between the thiol groups of cysteine residues by the process of oxidative folding.
Web Answer (1 Of 4):
Web cystine is composed of two cysteines linked by a disulfide bond (shown here in its neutral form). Web disulfide bond formation involves a reaction between the sulfhydryl (sh) side chains of two cysteine residues: Web what can form disulfide bonds? Web methionine residues as endogenous antioxidants in proteins.
An S− Anion From One Sulfhydryl Group Acts As A Nucleophile, Attacking The Side Chain Of A Second Cysteine To Create A Disulfide Bond, And In The Process Releases Electrons (Reducing Equivalents) For Transfer.
Thus methionine is more hydrophobic, sterically.