Why Don't Noble Gases Form Bonds
Why Don't Noble Gases Form Bonds - Given statement is that sulfur atom. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Web why do noble gases not form chemical bonds? Web noble gases already have a full outer valence shell.whcih can only hold a maximum of 8 electrons. Web the noble gases (group 18) are located in the far right of the periodic table and were previously referred to as the inert gases due to the fact that their filled. The full valence electron shells of these atoms make. The noble gasses already have a full valance shell so they don't need any more electrons. This tends to make them avoid other atoms because. Click to see the answer q: 10/26/2022 wiki user ∙ 11y ago study now see answer (1) best answer copy the compounds forming.
Noble gases are a group of. Web this group has been referred to as the “inert” gases, indicating that they are chemically inert, or unreactive. Web noble gases have a full valence shell, which is why they rarely form bonds with other atoms. Web noble gases elements are located in group 18 and known for their general electron configuration of n s 2 n p 6 ns^2 np^6 n s 2 n p 6 (with the exception of helium) which. Web why do noble gases not form chemical bonds? Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. The noble gasses already have a full valance shell so they don't need any more electrons. The noble gases are the least reactive of all the elements but the heavier ones do form some molecules. Web the noble gases (group 18) are located in the far right of the periodic table and were previously referred to as the inert gases due to the fact that their filled. Web noble gases already have a full outer valence shell.whcih can only hold a maximum of 8 electrons.
The noble gasses already have a full valance shell so they don't need any more electrons. Web why do noble gases not form chemical bonds? Web noble gases' outer shells are already filled with 8 electrons (other than he, which has 2, but is still filled and stable). Atoms bond with other elements to fill their outer shell, but they. Web noble gases already have a full outer valence shell.whcih can only hold a maximum of 8 electrons. Web noble gases do not form a chemical bond because their octet is complete and they do not have valence electrons. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Since the outer shell is filled, it doesn't have any empty electron spaces to. Noble gases are a group of. Web the noble gases (group 18) are located in the far right of the periodic table and were previously referred to as the inert gases due to the fact that their filled.
Noble gases What are the properties of noble gases? Noble gas
Web noble gases have a full valence shell, which is why they rarely form bonds with other atoms. 10/26/2022 wiki user ∙ 11y ago study now see answer (1) best answer copy the compounds forming. Click to see the answer q: Web why do noble gases not form chemical bonds? The noble gasses already have a full valance shell so.
Why Atoms Make Bonds Why Noble Gases are Stable Chemical Bonding
The full valence electron shells of these atoms make. Click to see the answer q: Helium and neon never form molecules. The clathrates , first described in 1949, [58]. Given statement is that sulfur atom.
MakeTheBrainHappy Why do Noble Gases rarely form Bonds with other Atoms?
Atoms bond with other elements to fill their outer shell, but they. The noble gasses already have a full valance shell so they don't need any more electrons. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. Web this group has been referred to as the “inert” gases, indicating that they are chemically inert,.
What Are Noble Gases? Definition and Properties
Atoms bond with other elements to fill their outer shell, but they. Another popular term is “noble gases,” suggesting that. Web noble gases elements are located in group 18 and known for their general electron configuration of n s 2 n p 6 ns^2 np^6 n s 2 n p 6 (with the exception of helium) which. Web why do.
Why do Noble Gases rarely form Bonds with other Atoms? MakeTheBrainHappy
Web noble gases' outer shells are already filled with 8 electrons (other than he, which has 2, but is still filled and stable). Since the outer shell is filled, it doesn't have any empty electron spaces to. Another popular term is “noble gases,” suggesting that. Web noble gases have a full valence shell, which is why they rarely form bonds.
Noble Gases; Gases, Inert; Rare Gases; Gases, Rare
Web noble gases already have a full outer valence shell.whcih can only hold a maximum of 8 electrons. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. The noble gasses already have a full valance shell so they don't need any more.
Why Don't Noble Gases Bond? Video & Lesson Transcript
Click to see the answer q: Web noble gases elements are located in group 18 and known for their general electron configuration of n s 2 n p 6 ns^2 np^6 n s 2 n p 6 (with the exception of helium) which. Web this group has been referred to as the “inert” gases, indicating that they are chemically inert,.
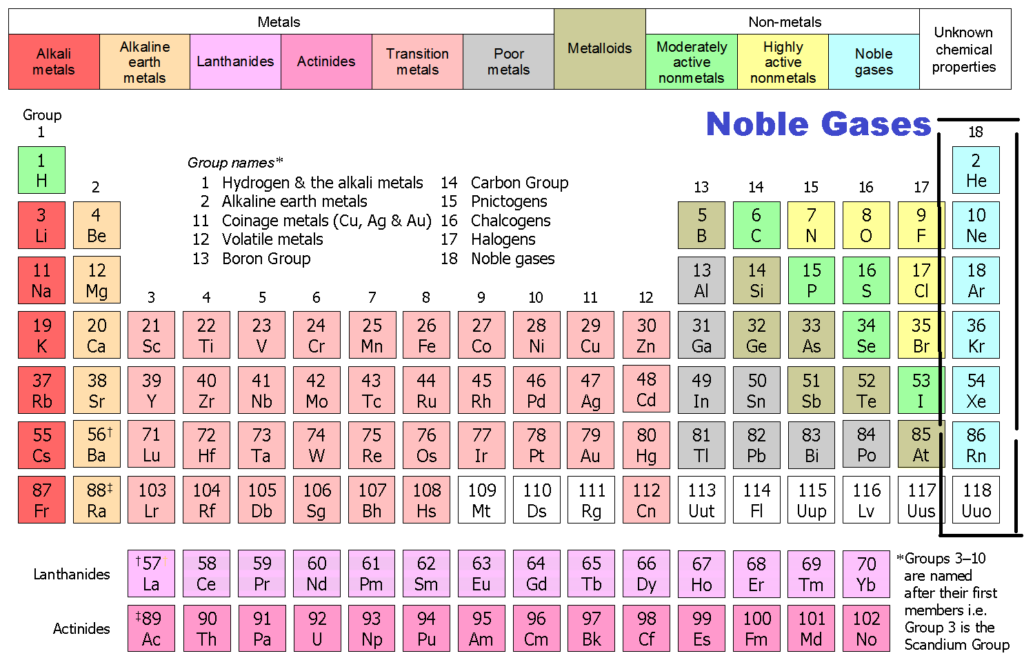
Group 18 The Noble Gases
Since the outer shell is filled, it doesn't have any empty electron spaces to. The clathrates , first described in 1949, [58]. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Atoms bond with other elements to fill their outer shell, but.
What Is The Reactivity Of Noble Gases howtogetalaid
Web noble gases already have a full outer valence shell.whcih can only hold a maximum of 8 electrons. Since the outer shell is filled, it doesn't have any empty electron spaces to. Web the noble gases (group 18) are located in the far right of the periodic table and were previously referred to as the inert gases due to the.
What Is The Reactivity Of Noble Gases howtogetalaid
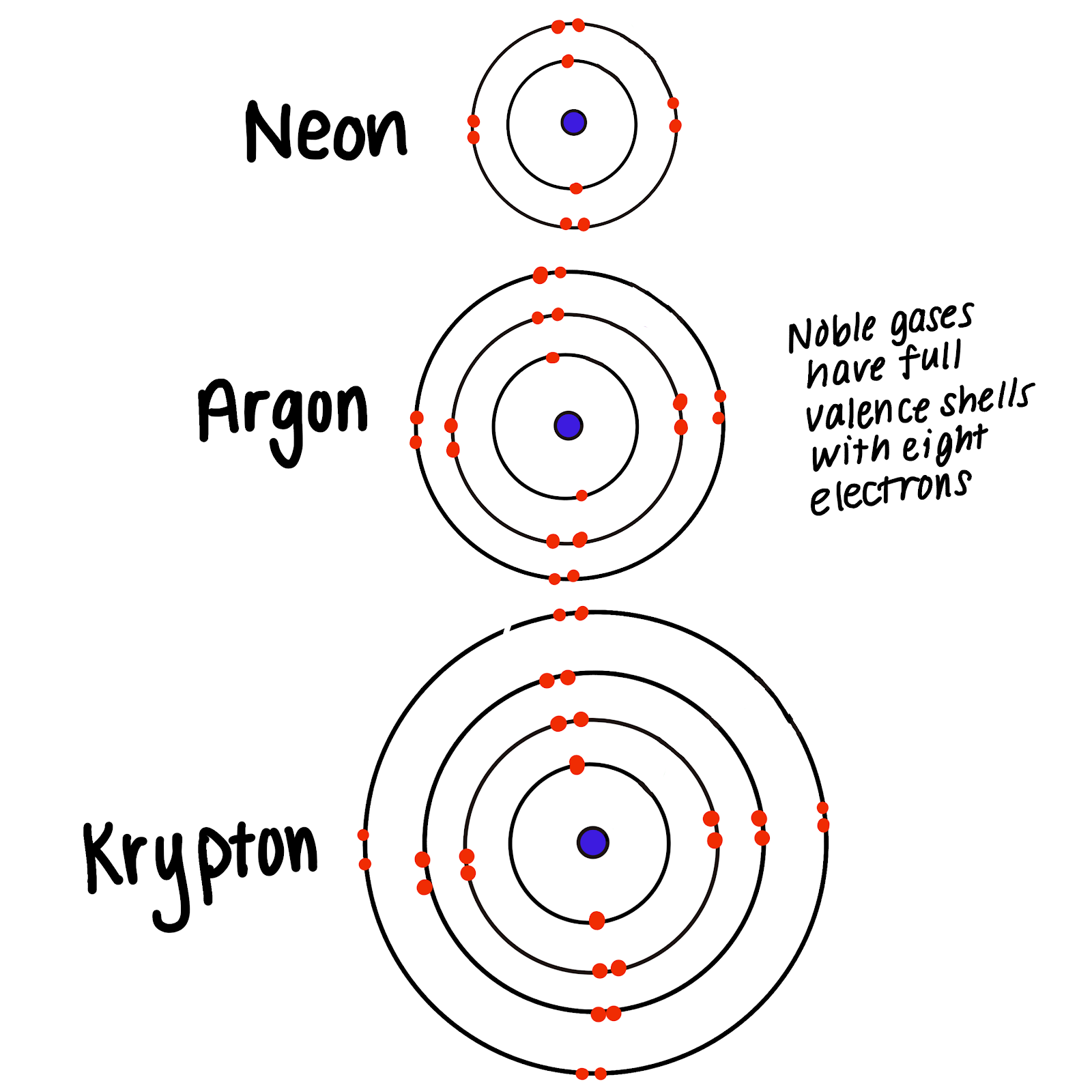
This graphic summarizes this quite well. Web noble gases have a full valence shell, which is why they rarely form bonds with other atoms. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. This tends to make them avoid other atoms because. Another popular term is “noble gases,” suggesting that.
Web Answer 1 Noble Gases Do Not Form A Chemical Bond Because Their Octet Is Complete And They Do Not Have Valence Electrons.
10/26/2022 wiki user ∙ 11y ago study now see answer (1) best answer copy the compounds forming. Web the noble gases (group 18) are located in the far right of the periodic table and were previously referred to as the inert gases due to the fact that their filled. Since they have full outer. Web this group has been referred to as the “inert” gases, indicating that they are chemically inert, or unreactive.
Web Noble Gases Are Odorless, Colorless, Nonflammable, And Monotonic Gases That Have Low Chemical Reactivity.
Web noble gases have a full valence shell, which is why they rarely form bonds with other atoms. Atoms bond with other elements to fill their outer shell, but they. This tends to make them avoid other atoms because. By gaining 2 electrons, a sulfur atom attains an electron arrangement like that of the noble gas… a:
Web Noble Gases Already Have A Full Outer Valence Shell.whcih Can Only Hold A Maximum Of 8 Electrons.
This graphic summarizes this quite well. The noble gasses already have a full valance shell so they don't need any more electrons. Web noble gases' outer shells are already filled with 8 electrons (other than he, which has 2, but is still filled and stable). Noble gases are a group of.
Another Popular Term Is “Noble Gases,” Suggesting That.
Web noble gases do not form a chemical bond because their octet is complete and they do not have valence electrons. The full valence electron shells of these atoms make. The noble gases are the least reactive of all the elements but the heavier ones do form some molecules. Since the outer shell is filled, it doesn't have any empty electron spaces to.






.PNG)